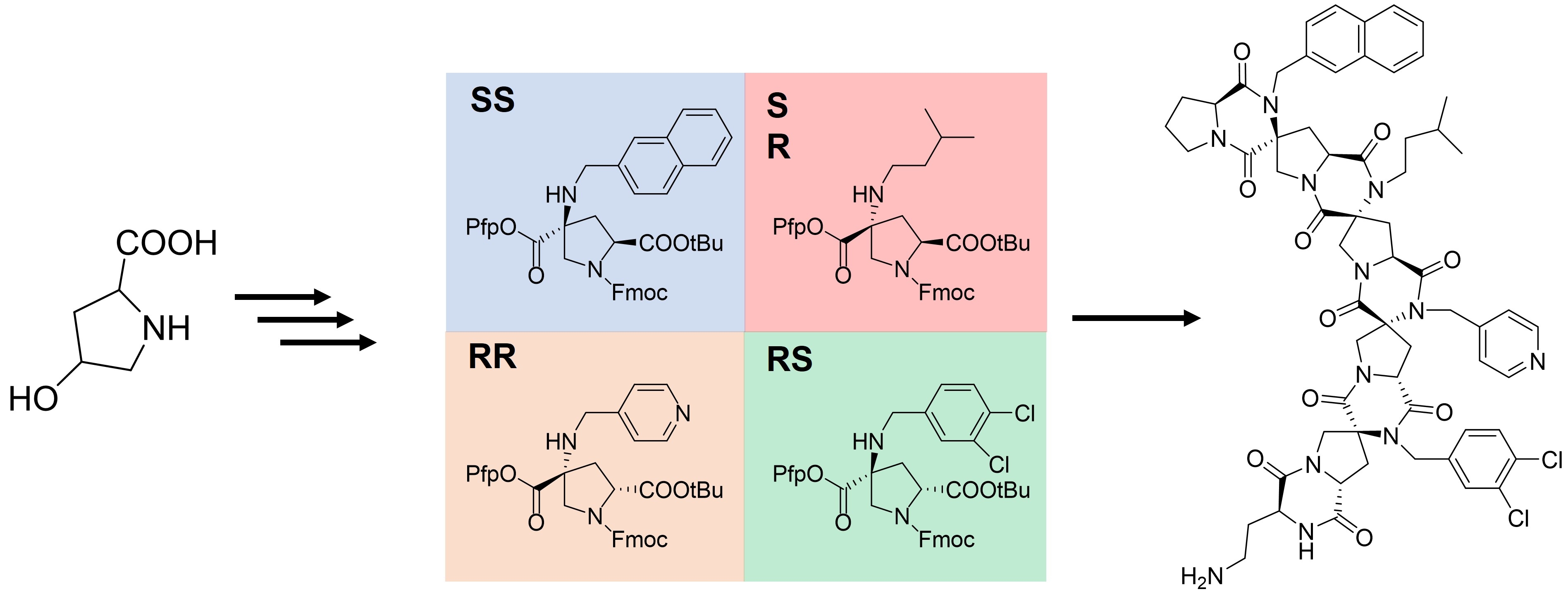
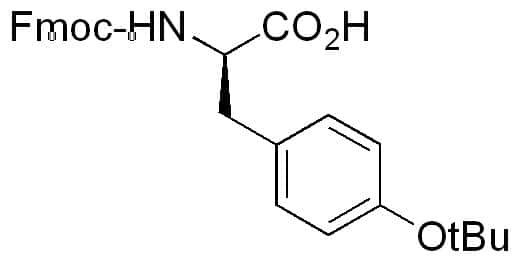
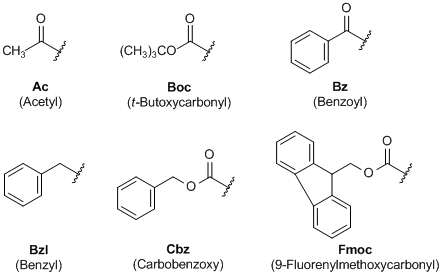
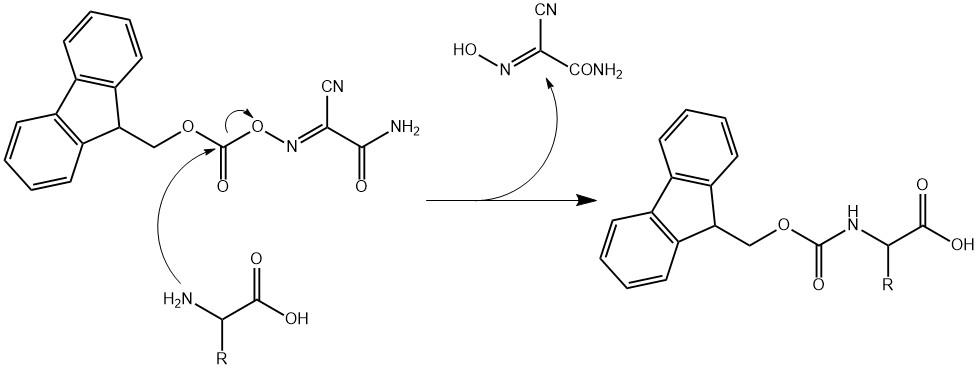
Large-Scale Syntheses of FMOC-Protected Non-Proteogenic Amino Acids: Useful Building Blocks for Combinatorial Libraries | Organic Process Research & Development

Nα‐Fmoc‐Protected ω‐Azido‐ and ω‐Alkynyl‐L‐amino Acids as Building Blocks for the Synthesis of “Clickable” Peptides - Le Chevalier Isaad - 2008 - European Journal of Organic Chemistry - Wiley Online Library

Development of Fmoc-Protected Bis-Amino Acids toward Automated Synthesis of Highly Functionalized Spiroligomers | Organic Letters

Synthesis of Fmoc-protected 4-N,N,-dimethylaminophthalimidoalanine (1)... | Download Scientific Diagram
Fmoc-L-Histidine-(Trityl), 100 g, CAS No. 109425-51-6 | Fluorenylmethylene / Fmoc | Amino acids, protected | Amino Acid Derivatives | Amino Acids and Amino Acid Derivatives | Organic & Bioorganic Chemicals | Chemicals | Carl Roth - International
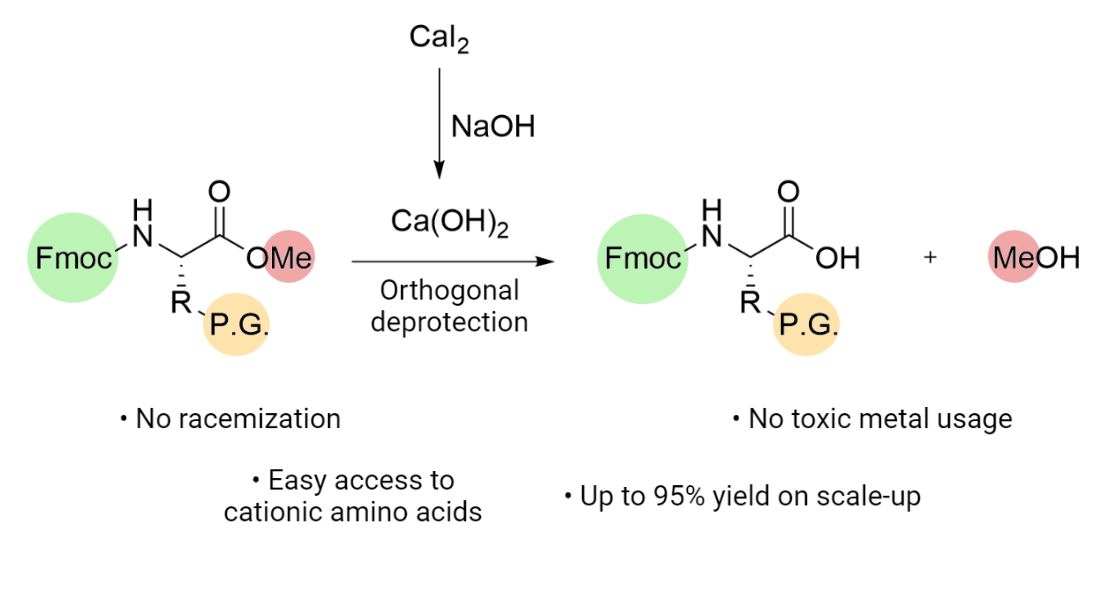
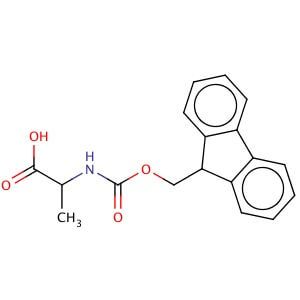
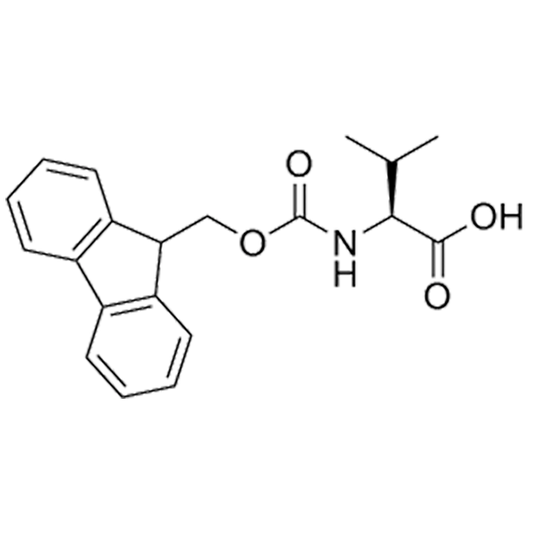
Molecules | Free Full-Text | Efficient Fmoc-Protected Amino Ester Hydrolysis Using Green Calcium(II) Iodide as a Protective Agent
![PDF] Fmoc-2-mercaptobenzothiazole, for the introduction of the Fmoc moiety free of side-reactions. | Semantic Scholar PDF] Fmoc-2-mercaptobenzothiazole, for the introduction of the Fmoc moiety free of side-reactions. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3232d408ed68af4d7d71eaef2557203a4a59a684/5-Figure2-1.png)
PDF] Fmoc-2-mercaptobenzothiazole, for the introduction of the Fmoc moiety free of side-reactions. | Semantic Scholar

An Fmoc protecting group can be removed from an amino acid by treatment with the amine base piperidine. Propose a mechanism. | Homework.Study.com