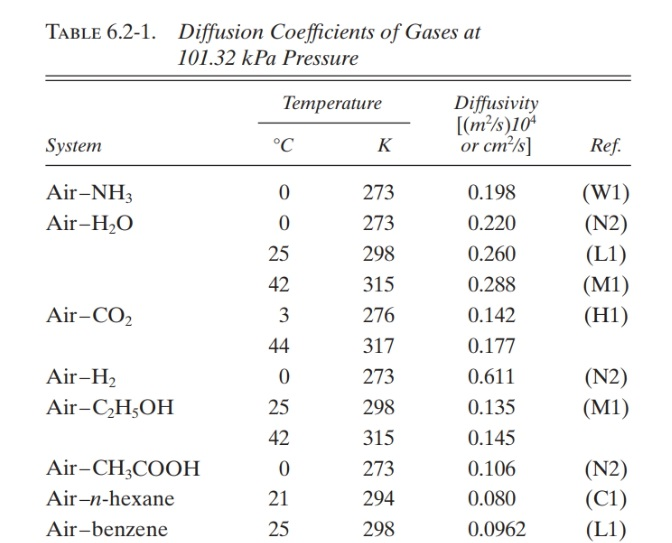
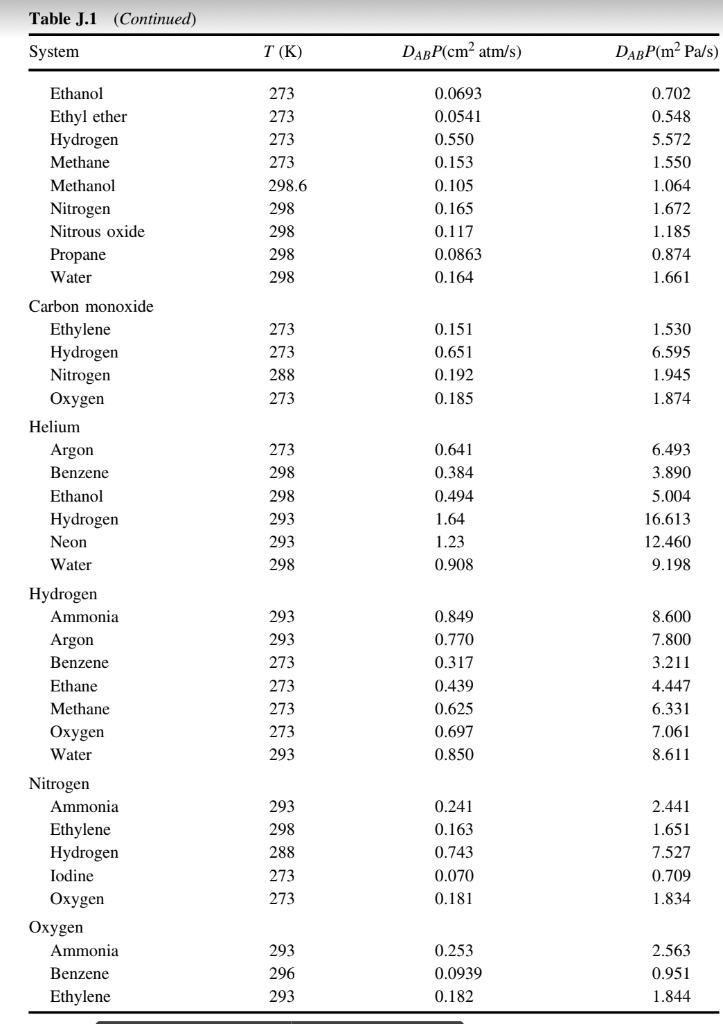
Investigation of tracer gas transport in a new numerical model of lung acini | Medical & Biological Engineering & Computing

Table 1 from Three-Porosity Model for Predicting the Gas Diffusion Coefficient in Undisturbed Soil | Semantic Scholar
![PDF] Self-Diffusion Coefficients of Lennard-Jones Liquids and Gases for Various Models in the Modified Free Volume Theory:Tables | Semantic Scholar PDF] Self-Diffusion Coefficients of Lennard-Jones Liquids and Gases for Various Models in the Modified Free Volume Theory:Tables | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f5cfa3309b95ccd3f1ea1910385f7eac8bbb212f/5-Table1-1.png)
PDF] Self-Diffusion Coefficients of Lennard-Jones Liquids and Gases for Various Models in the Modified Free Volume Theory:Tables | Semantic Scholar
Molecular diffusion coefficients of several gases, ions and molecules.... | Download Scientific Diagram

SOLVED: According to the Kinetic Theory of gases, the diffusion coefficient; D, of a gas can be described using the following equation: 1 D = v^ 3 Using this ratio calculates the
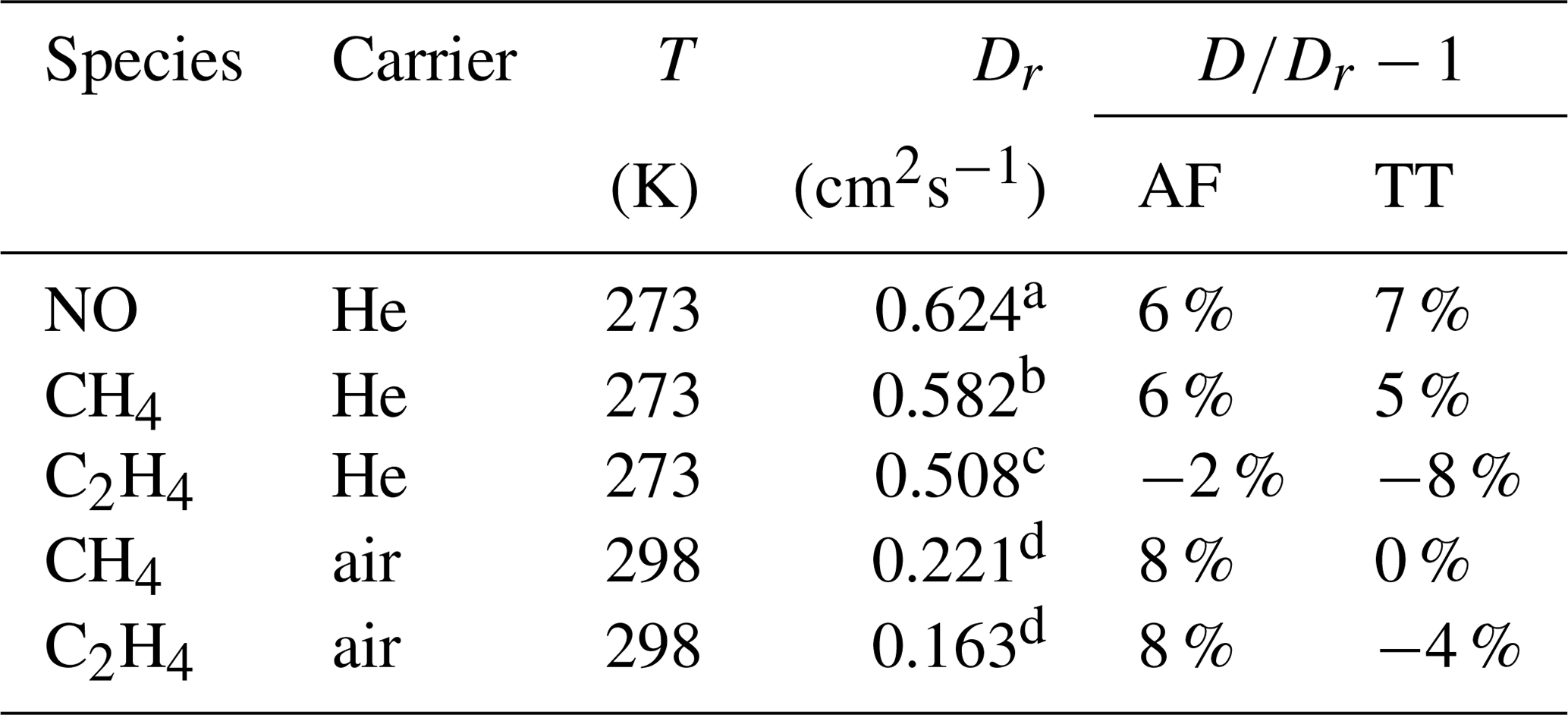
ACP - Technical note: Determination of binary gas-phase diffusion coefficients of unstable and adsorbing atmospheric trace gases at low temperature – arrested flow and twin tube method

Calculation of the diffusion coefficient of gas diffusion in coal: The comparison of numerical model and traditional analytical model - ScienceDirect
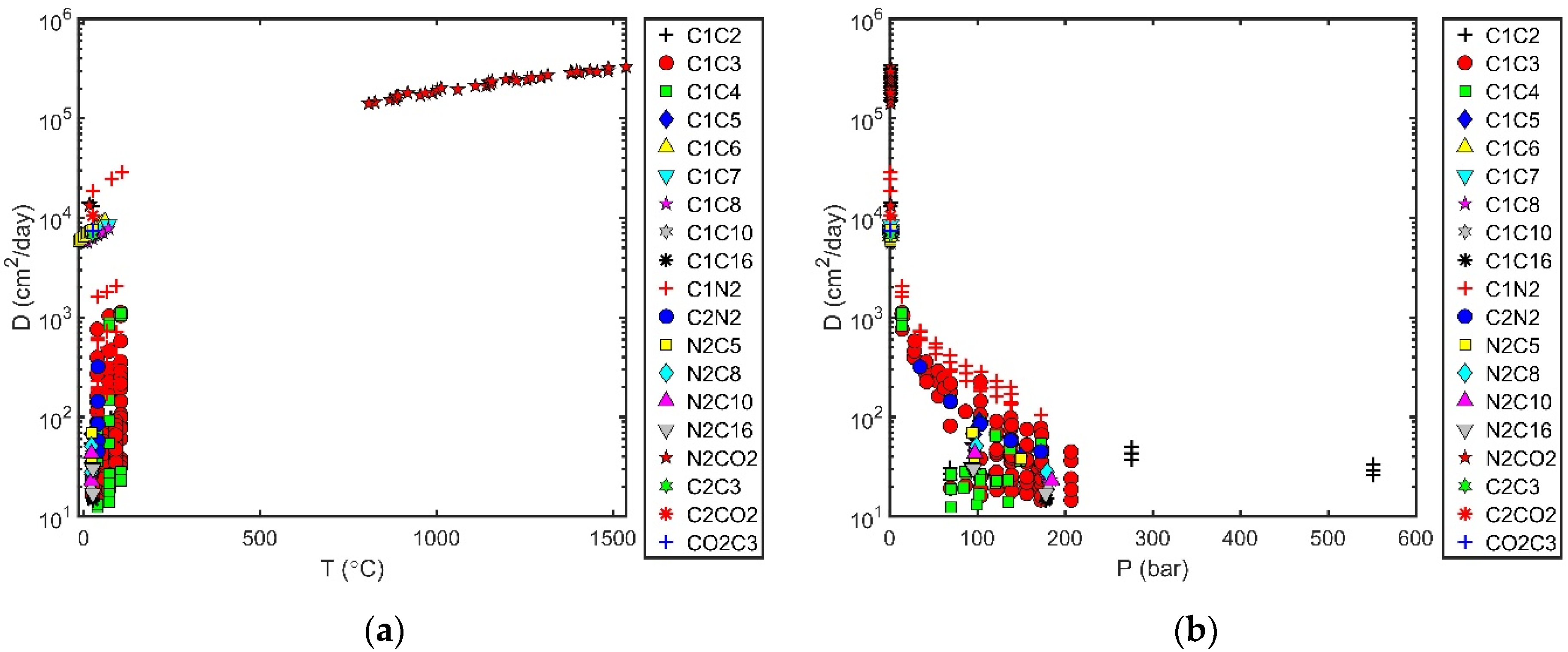
Processes | Free Full-Text | Diffusion Coefficients in Systems Related to Reservoir Fluids: Available Data and Evaluation of Correlations

The diffusion coefficient of an ideal gas is proportional to its mean path and mean speed. The absolute temperature of an ideal gas is increased 4 times and its pressure is increased

Diffusion Coefficients of CO2 and N2 in Water at Temperatures between 298.15 K and 423.15 K at Pressures up to 45 MPa | Journal of Chemical & Engineering Data

Molecules | Free Full-Text | Difference Analysis of Gas Molecules Diffusion Behavior in Natural Ester and Mineral Oil Based on Molecular Dynamic Simulation